|
Back to Blog
Ion bonding service5/8/2023  Owing to the fact that each element will have a different number of valence electrons, they can exhibit different number of valence states. The electronegativity of every element depends on the number of valence electrons it carries. These electrons are termed as valence electrons, and they play a key role in bond formation. Valence Electrons and ElectronegativityĪll the elements possess a certain charge, which is expressed as the number of electrons they carry in the outermost or valence shell of their orbit. Chemical bonds are of various types and possess varied strengths.ġ. Atoms of the same or different elements are combined by various chemical bonds in order to keep the molecules together and thus, they confer stability to the resulting compounds. 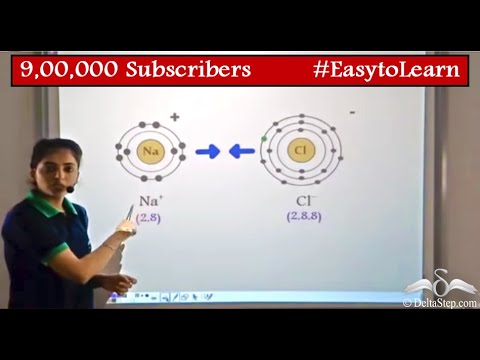
Talking about salt, have you ever wondered how sodium and chlorine combine to form a salt (sodium chloride)? Well, it is due to a process of bond formation known as ionic bonding.Īll the chemical compounds are formed due to various combinations of constituent elements. Accordingly, these substances tend to be hard and nonvolatile.Chemistry is fascinating, and it is present everywhere around us, be it the beautiful raindrops on your windowpane, sweet sugars in the fruits, or the salt that makes your food tastier. The magnitude of the electrostatic forces in ionic crystals is considerable. In short, the ions are so arranged that the positive and negative charges alternate and balance one another, the overall charge of the entire substance being zero. In ionic crystalline solids of this kind, the electrostatic forces of attraction between opposite charges and repulsion between similar charges orient the ions in such a manner that every positive ion becomes surrounded by negative ions and vice versa. Ionic bonding results in compounds known as ionic, or electrovalent, compounds, which are best exemplified by the compounds formed between nonmetals and the alkali and alkaline-earth metals. For full treatment, see chemical bonding: The formation of ionic bonds. A brief treatment of ionic bonds follows. The atom that loses the electrons becomes a positively charged ion ( cation), while the one that gains them becomes a negatively charged ion ( anion). Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom. Ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed

